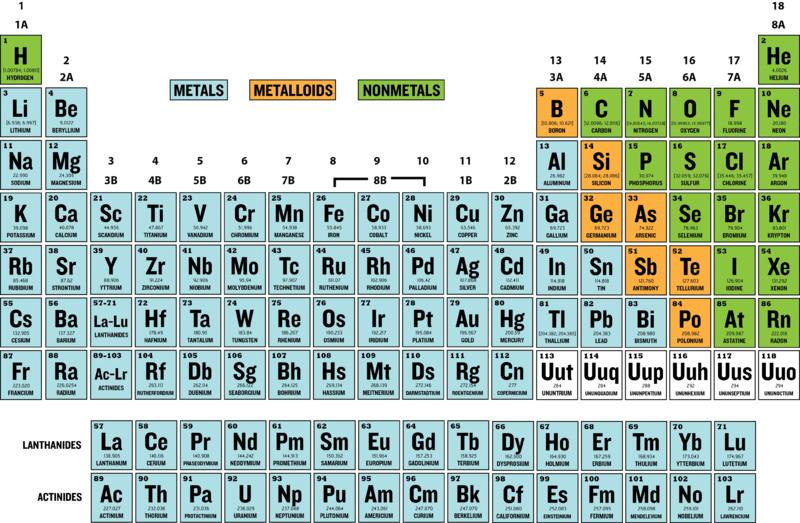
Periodic Table
Periods are the horizontal rows of elements. Each row has a specific number, which tell you how many energy levels each element in that row has. Energy levels are the invisible rings that contain electrons.
Groups, or families, are the vertical columns of elements. The number of each group tells you how many valence electrons are in each element. Valence electrons are electrons that can come and go from an atom, making it an ion. This determines how reactive an element is.
The number of valence electrons is represented by the group numbers. For example, the group number for sodium is 1, so it has 1 valence electron.
Metals cover most of the periodic table. Metals cover groups 1-13 with the exception of hydrogen and boron. The main metals are transition metals. Two other types of metals, lanthanides and actinides, were moved to the bottom of the table so the periodic table wouldn't be too wide.
Nonmetals are found on the upper right corner of the periodic table. This again is an exception to hydrogen, which was placed on the left because of its atomic number (one). Some nonmetals are also called noble gases.
Metalloids are located in the central area of the right side of the table. They shape up to be a diagonal line that separates metals from nonmetals. There are one to two metalloids in the first six rows and they are found in groups thirteen through sixteen.
The atomic number of an element tells us how many protons are in an element's atom. This technically gives us the number of electrons in an atom too, as long as it's stable. The atomic number can be found at the top of an elements box. The periodic table starts with the smallest atomic number and grows bigger from left to right all the way down to 118.
The atomic mass is found below an element's symbol. It tells us the number of protons and neutrons in an atom's nucleus. You can subtract the atomic number from the (rounded) atomic mass to find how many neutrons there are separately from protons.
Atoms always want to have their outer shell filled up. Elements on the very right have atoms that have their outer shell completely filled up with eight valence electrons. They are the least reactive. Elements on the very left only have one valence electron, so they just want to keep moving their electrons around. They are the most reactive. On the periodic table, the most reactive elements are on the left and they get less reactive the farther to the right you go.
Metals can be classified by if they are conductors (they let heat and electricity pass through them), and if they have big density. Also, they're malleable, which means they can be hammered into thin sheets and some are shiny and lustrous.
The properties of nonmetals are basically the opposite of metals. They are poor conductors, can't be hammered, and aren't shiny or lustrous. However, most nonmetals have the ability to gain electrons easily.
Metalloids have mixed properties, so they're hard to characterize. The physical appearance of a metalloid is that it's solid, brittle, has a metallic luster, and is semi-conductive. Chemically, they can form glasses and alloys with metals. Also, some metalloids contract on melting.
Groups, or families, are the vertical columns of elements. The number of each group tells you how many valence electrons are in each element. Valence electrons are electrons that can come and go from an atom, making it an ion. This determines how reactive an element is.
The number of valence electrons is represented by the group numbers. For example, the group number for sodium is 1, so it has 1 valence electron.
Metals cover most of the periodic table. Metals cover groups 1-13 with the exception of hydrogen and boron. The main metals are transition metals. Two other types of metals, lanthanides and actinides, were moved to the bottom of the table so the periodic table wouldn't be too wide.
Nonmetals are found on the upper right corner of the periodic table. This again is an exception to hydrogen, which was placed on the left because of its atomic number (one). Some nonmetals are also called noble gases.
Metalloids are located in the central area of the right side of the table. They shape up to be a diagonal line that separates metals from nonmetals. There are one to two metalloids in the first six rows and they are found in groups thirteen through sixteen.
The atomic number of an element tells us how many protons are in an element's atom. This technically gives us the number of electrons in an atom too, as long as it's stable. The atomic number can be found at the top of an elements box. The periodic table starts with the smallest atomic number and grows bigger from left to right all the way down to 118.
The atomic mass is found below an element's symbol. It tells us the number of protons and neutrons in an atom's nucleus. You can subtract the atomic number from the (rounded) atomic mass to find how many neutrons there are separately from protons.
Atoms always want to have their outer shell filled up. Elements on the very right have atoms that have their outer shell completely filled up with eight valence electrons. They are the least reactive. Elements on the very left only have one valence electron, so they just want to keep moving their electrons around. They are the most reactive. On the periodic table, the most reactive elements are on the left and they get less reactive the farther to the right you go.
Metals can be classified by if they are conductors (they let heat and electricity pass through them), and if they have big density. Also, they're malleable, which means they can be hammered into thin sheets and some are shiny and lustrous.
The properties of nonmetals are basically the opposite of metals. They are poor conductors, can't be hammered, and aren't shiny or lustrous. However, most nonmetals have the ability to gain electrons easily.
Metalloids have mixed properties, so they're hard to characterize. The physical appearance of a metalloid is that it's solid, brittle, has a metallic luster, and is semi-conductive. Chemically, they can form glasses and alloys with metals. Also, some metalloids contract on melting.